Research Surrounding miR-181a Published
By Camren Clouthier | June 25 2020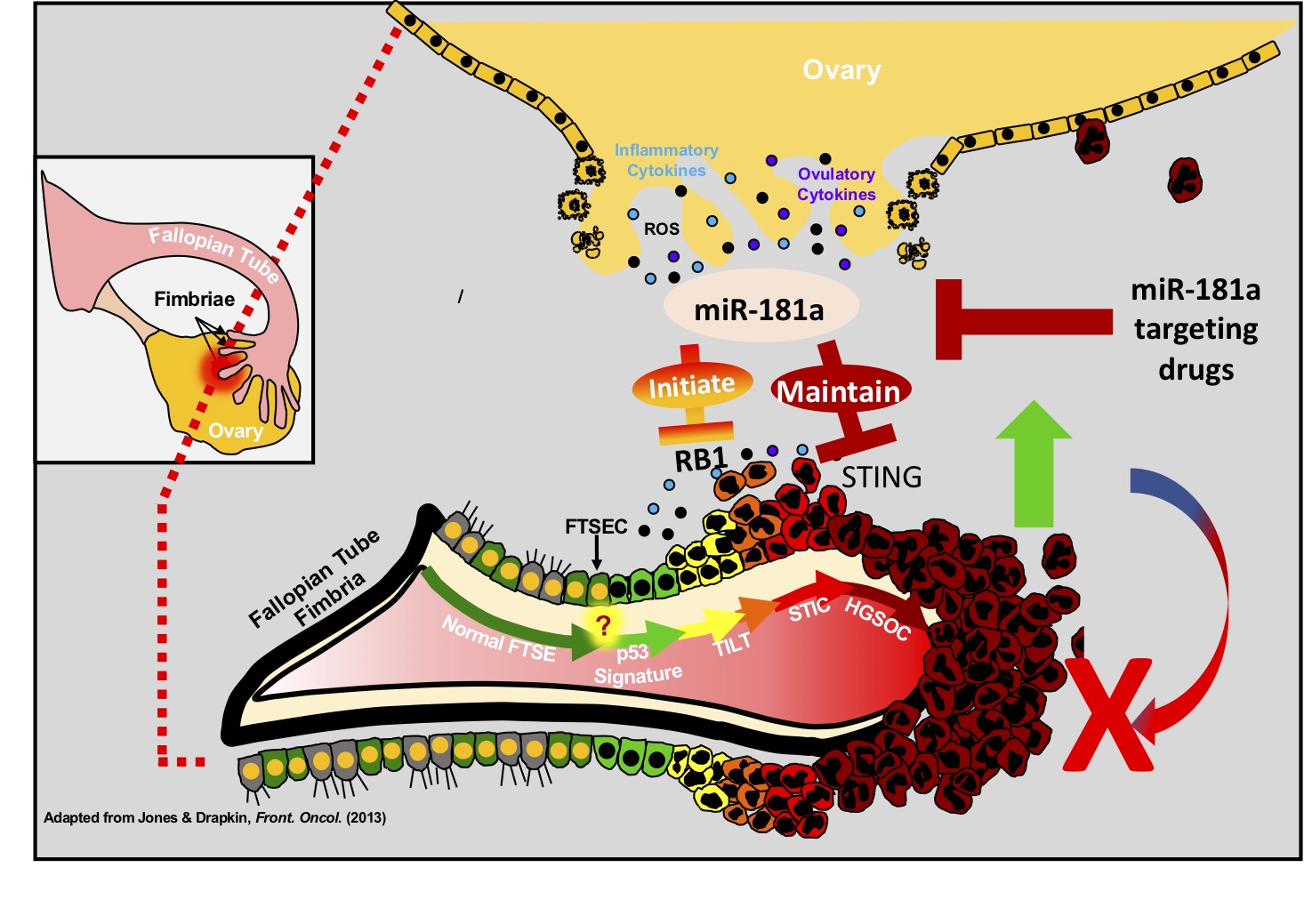 A new publication from the Department of Pathology and Dr. Analisa Difeo was just published in Nature Communications. The study focuses on miR-181a and how it initiates and perpetuates oncogenic transformation through the regulation of innate immune signaling.
A new publication from the Department of Pathology and Dr. Analisa Difeo was just published in Nature Communications. The study focuses on miR-181a and how it initiates and perpetuates oncogenic transformation through the regulation of innate immune signaling.
The research further examines how genomic instability (GI) predisposes cells to malignant transformation. However, the molecular mechanisms that allow for the propagation of cells with a high degree of genomic instability remain unclear. Here, experts report that miR-181a is able to transform fallopian tube secretory epithelial cells through the inhibition of RB1 and stimulator-of-interferon-genes (STING) to propagate cells with a high degree of GI. MiR-181a targeting of RB1 leads to profound nuclear defects and GI generating aberrant cytoplasmic DNA, however simultaneous miR-181a mediated inhibition of STING allows cells to bypass interferon-mediated cell death.
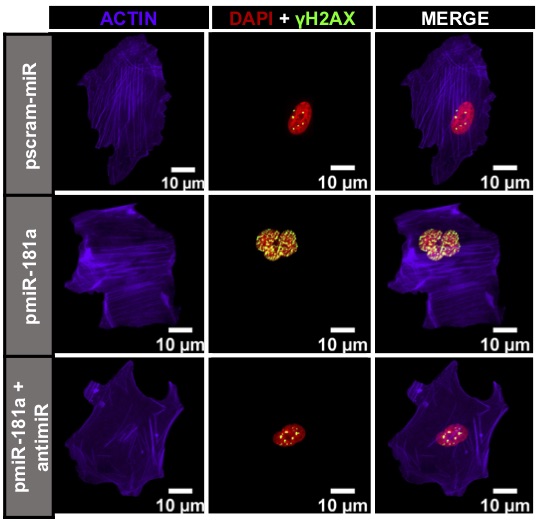 "We have found that miR-181a is the first oncomiR that is able to increase large scale genomic instability at the earliest stages of ovarian cancer development and through the downregulation of STING is able to suppress activation of the immunosurveillance machinery, and impair signaling in cancer cells creating a survival advantage," describes Dr. Difeo.
"We have found that miR-181a is the first oncomiR that is able to increase large scale genomic instability at the earliest stages of ovarian cancer development and through the downregulation of STING is able to suppress activation of the immunosurveillance machinery, and impair signaling in cancer cells creating a survival advantage," describes Dr. Difeo.
"We also found that miR-181a expression inversely correlated with STING expression and decreased immune cell infiltration/activation in ovarian cancer patient tumors. Thus, establishing miR-181a as a potential therapeutic target to sensitize tumors to immunotherapy or as a prognostic biomarker," notes Difeo.
Overall, the study uncovered that DNA oncoviruses are the only known inhibitors of STING that allow for cellular transformation, thus, our findings are the first to identify a miRNA that can downregulate STING expression to suppress activation of intrinsic interferon signaling. Finally, the study introduces miR-181a as a putative biomarker and identifies the miR-181a- STING axis as a promising target for therapeutic exploitation.
The full publication in Nature Communications is available here.
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
 ON THE COVER
ON THE COVER
